the viability of Mitra devices for therapeutic drug monitoring
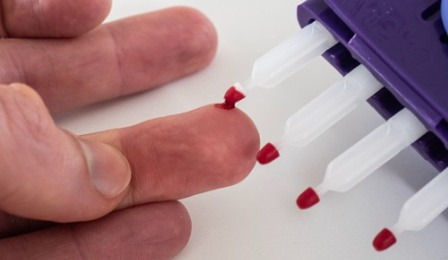

the viability of Mitra devices for therapeutic drug monitoring
Dec 16, 2024 1:32:11 PM
5
min read
urinary albumin creatinine ratio in dried microsamples


urinary albumin creatinine ratio in dried microsamples
Jun 3, 2024 9:00:00 AM
5
min read
Promega Publishes App Note on RNA Studies Using Mitra Devices


Promega Publishes App Note on RNA Studies Using Mitra Devices
Apr 18, 2024 3:44:28 PM
2
min read
comparing PEth and liver function enzymes to monitor alcohol abuse


comparing PEth and liver function enzymes to monitor alcohol abuse
Apr 1, 2024 9:00:00 AM
5
min read
the importance of clinical bridging for pharmacokinetic (PK) assays
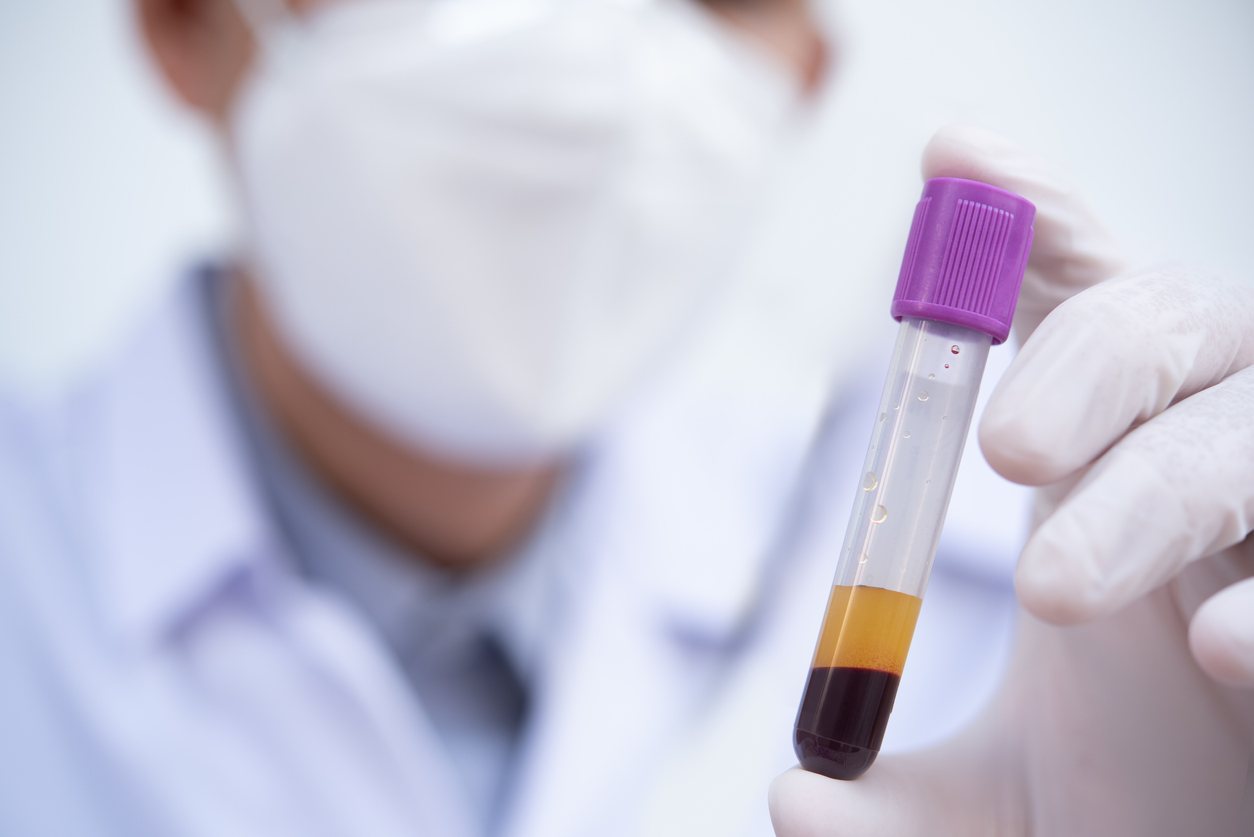

the importance of clinical bridging for pharmacokinetic (PK) assays
Mar 11, 2024 9:00:00 AM
6
min read
saliva microsampling to monitor oral fluid for epilepsy


saliva microsampling to monitor oral fluid for epilepsy
Feb 12, 2024 9:00:00 AM
5
min read
