Share this
Trajan’s Microsampling Devices Earn CE-IVD Registration in Europe, UK
by Neoteryx Microsampling on June 2,2023
June 2023 — Trajan Scientific and Medical, a leading provider of analytical and life sciences products and solutions, today announced the regulatory approval of its Neoteryx remote blood microsampling devices, the Mitra® and hemaPEN® for clinical use in Europe and the United Kingdom.
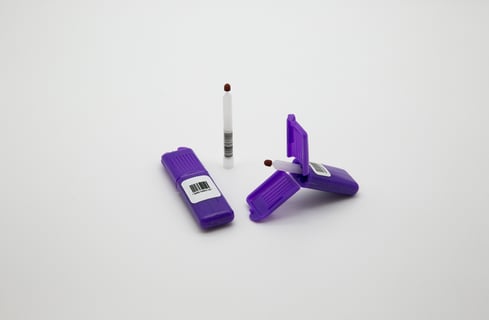 Trajan has successfully fulfilled the required product safety and performance requirements under the In Vitro Diagnostic Medical Devices Regulation (IVDR) in Europe and the Medicines and Healthcare Products Regulatory Agency (MHRA) in the United Kingdom.
Trajan has successfully fulfilled the required product safety and performance requirements under the In Vitro Diagnostic Medical Devices Regulation (IVDR) in Europe and the Medicines and Healthcare Products Regulatory Agency (MHRA) in the United Kingdom.
Having earned this CE-IVD regulatory registration, Trajan expands the capabilities of its Neoteryx® microsampling products to provide decentralized, person-centered, minimally invasive blood collection in both clinical and research settings.
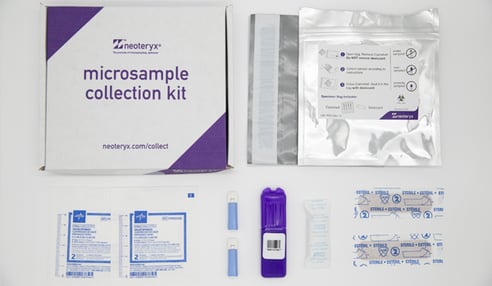 The Mitra device, based on volumetric absorptive microsampling with VAMS® technology, is a finger-stick blood collection solution for delivery of dried samples to the lab.
The Mitra device, based on volumetric absorptive microsampling with VAMS® technology, is a finger-stick blood collection solution for delivery of dried samples to the lab.
The hemaPEN device, based on volumetric microsampling with capillary and next-generation DBS technology, uses a single finger-stick to simultaneously collect 4 DBS blood samples of equal volume.
 Mitra and hemaPEN devices can be used remotely to collect blood samples through an easy, virtually painless process. These devices can be used to ship dried blood samples via standard mail, and they deliver precise volumes and high sample quality compared to traditional DBS or other remote blood sampling technologies.
Mitra and hemaPEN devices can be used remotely to collect blood samples through an easy, virtually painless process. These devices can be used to ship dried blood samples via standard mail, and they deliver precise volumes and high sample quality compared to traditional DBS or other remote blood sampling technologies.
The precision and quality of the microsamples have been validated through studies conducted by clinical labs, pharmaceutical companies, healthcare organizations, and academic institutions worldwide, showcasing their seamless integration into laboratory analysis workflows.
“The updated regulatory approval of Trajan’s blood microsampling devices in the UK and European Union is another step forward towards our vision of personalized, preventative data-based healthcare,” said Stephen Tomisich, CEO and founder, Trajan. “Our growing range of microsampling technologies allow almost anyone to take an analytically viable sample outside the clinical setting, anytime, anywhere. In our view, that is essential to enable the healthcare systems of the future.”
About Trajan Scientific and Medical
Trajan is a global developer and manufacturer of analytical and life sciences products and devices founded to enrich personal health through scientific tools and solutions. We aim to support science that benefits people. Trajan’s products and solutions are used in the analysis of biological, food, and environmental samples. Trajan has a portfolio and pipeline of new technologies which support the move towards decentralized personalized data-based healthcare.
Trajan comprises more than 680 people with seven manufacturing sites across the US, Australia, Europe, and Malaysia, and operations in Australia, the US, Asia, and Europe. Trajan’s products and solutions are marketed under multiple product brands, including Neoteryx, serving a range of industries in countries around the world. For more information, visit www.trajanscimed.com.
About Neoteryx, Microsampling Solutions by Trajan
Neoteryx, a world leader in microsampling devices, was acquired by Trajan in 2021. Today, Trajan’s full line of microsampling solutions carry the Neoteryx brand. The Neoteryx range of microsampling products provide precise, quantitative remote specimen collection. They are easy to use and amenable to automated analysis in the lab, easing workflows and enabling decentralized research and healthcare models.
The product portfolio includes the Mitra® device based on VAMS® technology and the hemaPEN® that advances both capillary and dried blood spot (DBS) technologies, with more microsampling products in the pipeline. For more information on Trajan’s Neoteryx range of microsampling solutions, visit www.neoteryx.com.
Explore published journal articles describing how researchers and clinical practitioners are applying microsampling in their industries. Visit our Microsampling Resource Library:
Image Credits: Trajan, Neoteryx
Share this
- Microsampling (41)
- Industry News, Microsampling News (37)
- Mitra® Device (34)
- Company Press Release, Product Press Release (22)
- Research, Remote Research (18)
- Infectious Disease, Vaccines, COVID-19 (15)
- Clinical Trials, Clinical Research (14)
- Biomonitoring, Health, Wellness (10)
- Blood Microsampling, Serology (10)
- Decentralized Clinical Trial (DCT) (9)
- Omics, Multi-Omics (7)
- Skin Microsampling, Microbiopsy (6)
- Venipuncture Alternative (6)
- Harpera Device (4)
- Specimen Collection (3)
- Toxicology, Doping, Drug/Alcohol Monitoring, PEth (3)
- Pharmaceuticals, Drug Development (2)
- Therapeutic Drug Monitoring, TDM (2)
- Antibodies, MAbs (1)
- Environmental Toxins, Exposures (1)
- Preclinical Research, Animal Studies (1)
- hemaPEN® Device (1)
- April 2026 (1)
- March 2026 (1)
- February 2026 (1)
- January 2026 (1)
- December 2025 (1)
- May 2025 (1)
- April 2025 (1)
- February 2025 (4)
- September 2024 (1)
- August 2024 (1)
- May 2024 (2)
- January 2024 (1)
- December 2023 (2)
- November 2023 (3)
- October 2023 (2)
- September 2023 (1)
- August 2023 (3)
- July 2023 (3)
- June 2023 (1)
- May 2023 (2)
- April 2023 (1)
- March 2023 (2)
- February 2023 (1)
- January 2023 (2)
- December 2022 (1)
- November 2022 (1)
- October 2022 (2)
- August 2022 (1)
- April 2022 (1)
- February 2022 (1)
- January 2022 (1)
- December 2021 (1)
- November 2021 (1)
- October 2021 (2)
- September 2021 (1)
- August 2021 (2)
- July 2021 (2)
- June 2021 (2)
- April 2021 (1)
- March 2021 (2)
- February 2021 (1)
- January 2021 (1)
- December 2020 (1)
- November 2020 (1)
- October 2020 (1)
- September 2020 (2)
- August 2020 (3)
- July 2020 (3)
- June 2020 (2)
- May 2020 (1)
- April 2020 (3)
- October 2019 (1)
- March 2019 (1)
- January 2019 (1)
- November 2018 (1)
- August 2018 (1)
- July 2018 (1)
- June 2017 (1)
- April 2017 (1)
- March 2017 (1)
- February 2017 (1)
- May 2016 (1)
- December 2015 (1)
- October 2015 (1)
- August 2015 (1)
- August 2014 (1)
- July 2014 (1)



No Comments Yet
Let us know what you think