Share this
pediatric pk studies under pressure: DBS vs. VAMS in Côte d’Ivoire
by Neoteryx Microsampling on Apr 2, 2018 4:44:00 AM
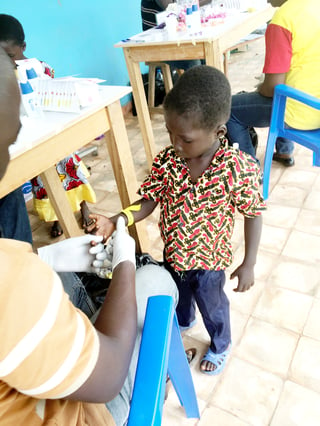 Scientists and researchers from the Swiss Tropical and Public Health Institute recently provided new, strong evidence for the efficacy of pediatric pharmacokinetic (PK) studies using Mitra® devices.
Scientists and researchers from the Swiss Tropical and Public Health Institute recently provided new, strong evidence for the efficacy of pediatric pharmacokinetic (PK) studies using Mitra® devices.
They tested Mitra technology against standard-issue DBS cards, within the framework of a randomized controlled trial, on 35 school-aged children in rural Côte d’Ivoire infected with Schistosoma haematobium.
For those who have been curious about the capabilities of microsampling technology in areas with limited access to standard laboratory facilities, this is exciting news indeed.
Thanks to significant scientific and technological advances, it is now possible to collect a large amount of information from a small amount of biological fluid. This allows for the elimination of cold-chain shipping and storage, among other bulky and expensive requirements of wet blood collection, and thus opens new frontiers for blood collection in low-resourced regions. However, this process has long been stymied by the limitations of common DBS technology, chief among them the “hematocrit effect,” which hamper the reliability of resultant data.
Mitra devices, based on Volumetric Absorptive Microsamping technology, are changing all this. They are designed to collect volumetrically accurate samples, thus eliminating the primary drawbacks formerly associated with dried blood specimen collection. To Jennifer Keiser, Ph.D., at the Swiss TPH, this innovation is of special interest.
A pharmacist by training, Keiser has a professorship in neglected tropical diseases. With the Swiss TPH, she has been involved in research in Africa for many years. “The Centre Suisse de Recherches Scientifiques in Abidjan is one of our main partner institutions,” says Keiser. “One of my postdocs is Ivorian and based in Abidjan.”
The Swiss TPH’s Mitra study stems from its ongoing work in the Côte d’Ivoire area. “I started my research group about twelve years ago,” says Keiser. “Since then, we are trying to improve treatment options for helminthiases, mainly schistosomiasis and soil-transmitted helminthiasis. We work from bench to field. I love the interdisciplinarity nature of the work and that our findings might result in policy changes, and in improved treatment options.”
For this project, the Swiss TPH scientists developed, optimized, and validated an extraction method for the study drug, praziquantel (PZQ), from VAMS® tips on Mitra devices. Using liquid chromatography-tandem mass spectrometry, they measured the analytes R- and S-praziquantel (R-/SPZQ) and the main human metabolite, R-trans-4-OH-praziquantel. Then, they compared results with Bland-Altman analysis to determine agreement between matrices.
For the Swiss TPH, this project required significant fieldwork, including screening stool of the study population for Schistosoma mansoni eggs and assessing infection intensity, performing physical and medical examination of infected children, proceeding with treatment with the study drug, and monitoring pharmacokinetics of same.
The results are impressive – from the perspective of microsampling, the study was a hands-down success. Strong positive correlation (R2 > 0.98) and agreement were observed between both matrices, but Mitra devices with VAMS tips revealed higher concentrations of all analytes in most patient samples compared to DBS: 63% for RPZQ, 49% for SPZQ, and 78% for the metabolite.
What’s more, Keiser's team found Mitra devices easier to use, too.
“The nurses taking the blood samples preferred this method over the DBS-method,” says Keiser. “Mitra doesn't need a ‘drying period,’ because the packaging is designed to not touch anything. Therefore, the Mitra stick can be put away immediately. DBS cards need to dry, which is in tropical area in not that fast. Having a couple of hundred cards standing around for drying is inconvenient, especially if working space is limited in the field and the DBS are exposed to sand, dirt, and wind.”
This is in keeping with the overall improved user experience of Mitra devices and Volumetric Absorptive Microsamping technology. The goal is to improve patient experience (which in turn promotes adherence, compliance, and subject retention), and at the same time create a better experience for providers, practitioners, and professionals.
If it can happen in some of the most underserved places on the globe, the world is truly and open landscape for this innovative, transformative sampling solution.

Share this
- Microsampling (206)
- Research, Remote Research (119)
- Venipuncture Alternative (105)
- Clinical Trials, Clinical Research (83)
- Mitra® Device (73)
- Therapeutic Drug Monitoring, TDM (51)
- Dried Blood Spot, DBS (39)
- Biomonitoring, Health, Wellness (30)
- Infectious Disease, Vaccines, COVID-19 (24)
- Blood Microsampling, Serology (23)
- Omics, Multi-Omics (21)
- Decentralized Clinical Trial (DCT) (20)
- Specimen Collection (18)
- Toxicology, Doping, Drug/Alcohol Monitoring, PEth (17)
- Skin Microsampling, Microbiopsy (14)
- hemaPEN® Device (13)
- Preclinical Research, Animal Studies (12)
- Pharmaceuticals, Drug Development (9)
- Harpera Device (7)
- Industry News, Microsampling News (5)
- Antibodies, MAbs (3)
- Company Press Release, Product Press Release (3)
- Environmental Toxins, Exposures (1)
- July 2025 (1)
- May 2025 (1)
- April 2025 (2)
- December 2024 (2)
- November 2024 (1)
- October 2024 (3)
- September 2024 (1)
- June 2024 (1)
- May 2024 (1)
- April 2024 (4)
- March 2024 (1)
- February 2024 (2)
- January 2024 (4)
- December 2023 (3)
- November 2023 (3)
- October 2023 (3)
- September 2023 (3)
- July 2023 (3)
- June 2023 (2)
- April 2023 (2)
- March 2023 (2)
- February 2023 (2)
- January 2023 (3)
- December 2022 (2)
- November 2022 (3)
- October 2022 (4)
- September 2022 (3)
- August 2022 (5)
- July 2022 (2)
- June 2022 (2)
- May 2022 (4)
- April 2022 (3)
- March 2022 (3)
- February 2022 (4)
- January 2022 (5)
- December 2021 (3)
- November 2021 (5)
- October 2021 (3)
- September 2021 (3)
- August 2021 (4)
- July 2021 (4)
- June 2021 (4)
- May 2021 (4)
- April 2021 (3)
- March 2021 (5)
- February 2021 (4)
- January 2021 (4)
- December 2020 (3)
- November 2020 (5)
- October 2020 (4)
- September 2020 (3)
- August 2020 (3)
- July 2020 (6)
- June 2020 (4)
- May 2020 (4)
- April 2020 (3)
- March 2020 (6)
- February 2020 (3)
- January 2020 (4)
- December 2019 (5)
- November 2019 (4)
- October 2019 (2)
- September 2019 (4)
- August 2019 (4)
- July 2019 (3)
- June 2019 (7)
- May 2019 (6)
- April 2019 (5)
- March 2019 (6)
- February 2019 (5)
- January 2019 (8)
- December 2018 (3)
- November 2018 (4)
- October 2018 (7)
- September 2018 (6)
- August 2018 (5)
- July 2018 (8)
- June 2018 (6)
- May 2018 (5)
- April 2018 (6)
- March 2018 (4)
- February 2018 (6)
- January 2018 (4)
- December 2017 (2)
- November 2017 (3)
- October 2017 (2)
- September 2017 (4)
- August 2017 (2)
- July 2017 (4)
- June 2017 (5)
- May 2017 (6)
- April 2017 (6)
- March 2017 (5)
- February 2017 (4)
- January 2017 (1)
- July 2016 (3)
- May 2016 (1)
- April 2016 (2)


Comments (1)