Share this
fast, accurate and remote tacrolimus blood level monitoring
by Neoteryx Microsampling on Jul 11, 2019 7:11:00 AM
At the 2019 meeting of the American Transplant Congress in Boston, scientists from the Oslo University Hospital, Rikshospitalet presented exciting findings regarding more patient-centered AUC-monitoring of tacrolimus, a cornerstone immunosuppressant, using remote blood microsampling approaches enabled by Mitra® devices based on VAMS® technology.
Researcher and PhD student Marte Theie Gustavsen (aka: Marte Theie Mol) gave a presentation on using Mitra with volumetric absorptive microsampling for therapeutic drug monitoring, or TDM, of patients who have had kidney transplants. After her presentation, she made time to answer a few questions about using remote microsampling for TDM. 
Tacrolimus Monitoring: Why It Matters
In the administration of tacrolimus, an immunosuppressant drug given to kidney transplant patients to help them avoid rejection, precise tacrolimus levels are required in the bloodstream. However, it can be challenging to get precise drug levels from blood samples.
Thus, more precise, more convenient methods of tacrolimus monitoring are in demand. It is important that monitoring is accurate, yet patients don't always comply with monitoring or properly adhere to their treatment, which can lead to negative effects of tacrolimus and other health problems.
“Too high exposure of tacrolimus increases the risk of hypertension, post-transplant diabetes mellitus, and neurotoxicity, while acute rejection episodes and development of donor specific antibodies are associated with too low exposure.”
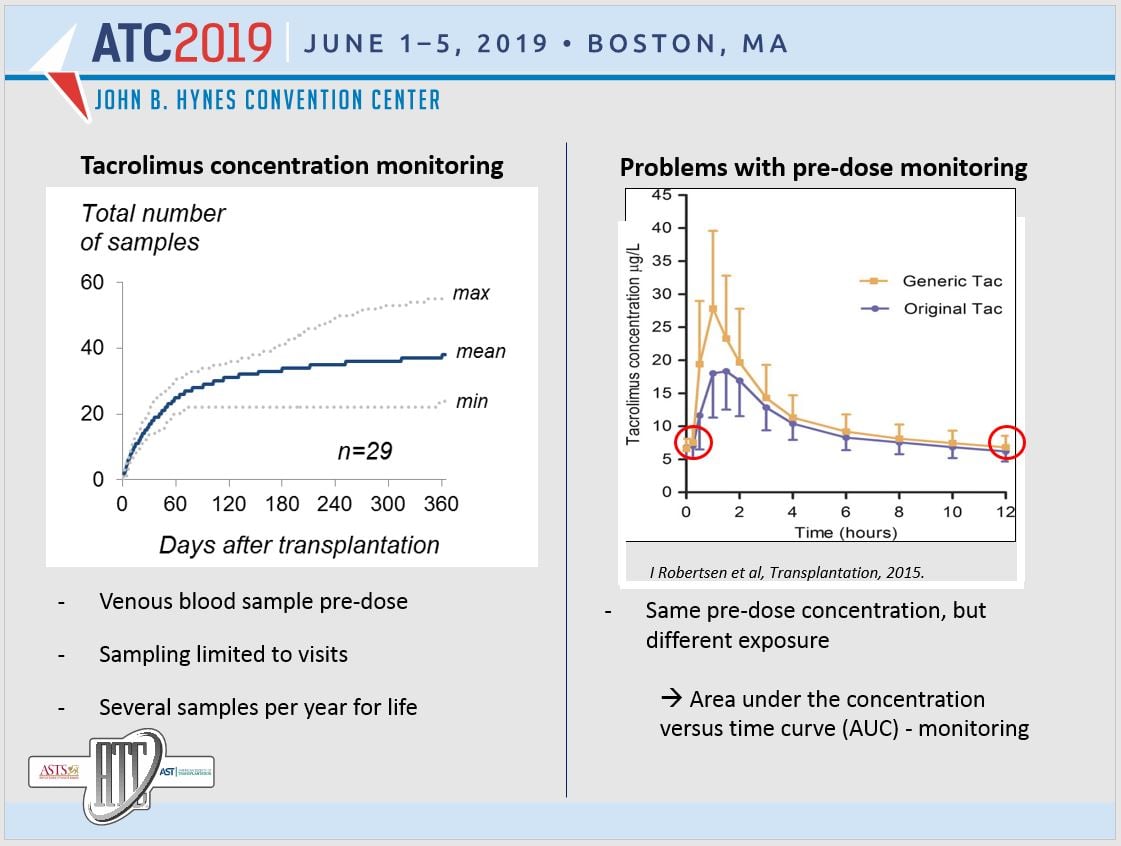
Tacrolimus Monitoring Research and Results
During their research, the Oslo team performed a 12-hour pharmacokinetic investigational study in 27 renal transplant patients. Blood sampling occurred 13 times during the dose interval, using both standard venipuncture blood collection and microsampling (finger-stick blood collection).
The Mitra microsample assay fulfilled the criteria on bioanalytical method validation, producing results comparable to those obtained through standard venipuncture. The team published its full results for further study. Their influence on tacrolimus monitoring is likely to be substantial.
Microsampling for Better Tacrolimus Monitoring
“Therapeutic drug monitoring of Tacrolimus is usually performed using pre-dose concentrations, mostly due to practical reasons,” says Gustavsen. “However, a pre-dose concentration gives little information about systemic exposure, and area under the concentration versus time curve - AUC - has been suggested to be the best pharmacokinetic marker for therapeutic drug monitoring of tacrolimus.” So how do we measure it?
“With the use of population pharmacokinetic models and limited sampling strategies, three optimally timed sampling points can give accurate predictions of AUC,” Gustavsen says. “By using microsampling, a sufficient number of blood samples within a dose interval, without any restriction in sampling time, can be obtained in an at-home setting by patients themselves. This will enable and simplify dose adjustments of tacrolimus based on AUC, which in turn hopefully will improve clinical outcome.”
The Oslo study showcases the convenience and user-friendliness of remote microsampling devices, both at home and in the lab, and why the remote approach to therapeutic drug monitoring is effective in keeping transplant patients healthy and safe at home, yet on the right track for post-transplant recovery and maintenance.
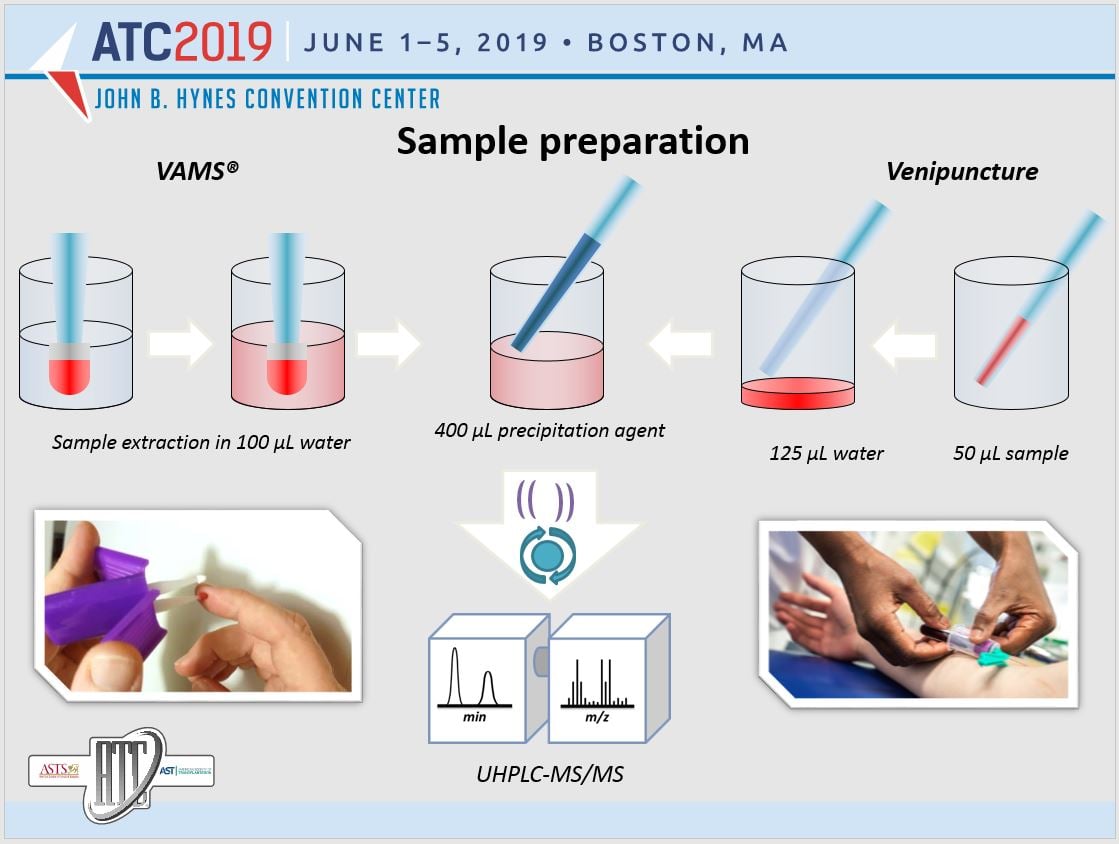
Goal: Best Possible Treatment for Transplant Patients
The Oslo team found that the use of remote blood microsampling devices made the study run much more smoothly than anticipated. “After systematic patient training and guidance on the microsample procedure and handling, the patients collected several microsamples by themselves in a home setting. None of the patients reported any sampling difficulties, which indicates that the clinical application of Mitra microsampling is possible,” says Gustavsen.
“The use of microsampling in at-home settings by patients themselves would simplify and improve the follow-up of renal transplant patients,” Gustavsen says. “This is a more patient-friendly way to obtain blood samples and will hopefully impact patients' quality of life for the better. After all, our goal is to give the best possible treatment to our patients.”
Thinking Long-term: Remote Patient Monitoring
“Renal transplant patients need lifelong follow-up with routinely blood sampling performed every month for the rest of their lives,” says Gustavsen. “Patients then have to come to the hospital in the early morning, draw a large blood volume, before waiting several hours to meet the physician. This is time-consuming, and some patients have to travel long distances.
Remote patient monitoring through remote microsampling paired with telehealth technologies would reduce patient burden and make the blood sample collection procedure more patient-friendly. A fixed sample volume from a single finger-prick can be obtained in a home setting. Compared to other home-based methods, blood volume absorbed on the VAMS tip ot a Mitra device is not dependent on hematocrit level, which is important, as the hematocrit level changes with time after transplantation.”
Thus, blood microsampling using Mitra devices with volumetric absorptive microsampling technology creates better long-term conditions for transplant patients. Meanwhile, it opens new opportunities and pathways of care across the landscape of immunosuppressant monitoring and patient-centered medicine.
“Patients were very motivated to participate in our study and saw the benefit this would have for them in the future,” says Gustavsen.
To learn more about using remote microsampling for your TDM programs, visit our page dedicated to: 
Image Credits: The Oslo University Hospital, Rikshospitalet; Trajan, Neoteryx

Share this
- Microsampling (206)
- Research, Remote Research (119)
- Venipuncture Alternative (105)
- Clinical Trials, Clinical Research (83)
- Mitra® Device (73)
- Therapeutic Drug Monitoring, TDM (51)
- Dried Blood Spot, DBS (39)
- Biomonitoring, Health, Wellness (30)
- Infectious Disease, Vaccines, COVID-19 (24)
- Blood Microsampling, Serology (23)
- Omics, Multi-Omics (21)
- Decentralized Clinical Trial (DCT) (20)
- Specimen Collection (18)
- Toxicology, Doping, Drug/Alcohol Monitoring, PEth (17)
- Skin Microsampling, Microbiopsy (14)
- hemaPEN® Device (13)
- Preclinical Research, Animal Studies (12)
- Pharmaceuticals, Drug Development (9)
- Harpera Device (7)
- Industry News, Microsampling News (5)
- Antibodies, MAbs (3)
- Company Press Release, Product Press Release (3)
- Environmental Toxins, Exposures (1)
- July 2025 (1)
- May 2025 (1)
- April 2025 (2)
- December 2024 (2)
- November 2024 (1)
- October 2024 (3)
- September 2024 (1)
- June 2024 (1)
- May 2024 (1)
- April 2024 (4)
- March 2024 (1)
- February 2024 (2)
- January 2024 (4)
- December 2023 (3)
- November 2023 (3)
- October 2023 (3)
- September 2023 (3)
- July 2023 (3)
- June 2023 (2)
- April 2023 (2)
- March 2023 (2)
- February 2023 (2)
- January 2023 (3)
- December 2022 (2)
- November 2022 (3)
- October 2022 (4)
- September 2022 (3)
- August 2022 (5)
- July 2022 (2)
- June 2022 (2)
- May 2022 (4)
- April 2022 (3)
- March 2022 (3)
- February 2022 (4)
- January 2022 (5)
- December 2021 (3)
- November 2021 (5)
- October 2021 (3)
- September 2021 (3)
- August 2021 (4)
- July 2021 (4)
- June 2021 (4)
- May 2021 (4)
- April 2021 (3)
- March 2021 (5)
- February 2021 (4)
- January 2021 (4)
- December 2020 (3)
- November 2020 (5)
- October 2020 (4)
- September 2020 (3)
- August 2020 (3)
- July 2020 (6)
- June 2020 (4)
- May 2020 (4)
- April 2020 (3)
- March 2020 (6)
- February 2020 (3)
- January 2020 (4)
- December 2019 (5)
- November 2019 (4)
- October 2019 (2)
- September 2019 (4)
- August 2019 (4)
- July 2019 (3)
- June 2019 (7)
- May 2019 (6)
- April 2019 (5)
- March 2019 (6)
- February 2019 (5)
- January 2019 (8)
- December 2018 (3)
- November 2018 (4)
- October 2018 (7)
- September 2018 (6)
- August 2018 (5)
- July 2018 (8)
- June 2018 (6)
- May 2018 (5)
- April 2018 (6)
- March 2018 (4)
- February 2018 (6)
- January 2018 (4)
- December 2017 (2)
- November 2017 (3)
- October 2017 (2)
- September 2017 (4)
- August 2017 (2)
- July 2017 (4)
- June 2017 (5)
- May 2017 (6)
- April 2017 (6)
- March 2017 (5)
- February 2017 (4)
- January 2017 (1)
- July 2016 (3)
- May 2016 (1)
- April 2016 (2)


Comments (2)