Share this
a breakthrough in capillary blood sampling for multi-omics studies
by James Rudge, PhD, Technical Director, Trajan on Mar 27, 2023 9:00:00 AM
Part 2: Using Blood Microsamples & Wearables to Profile Health Changes
In part one of this blog series, we introduced the pioneering microsampling and omics work of Michael Snyder, PhD, and his research team in the Snyder Lab at Stanford University. We outlined how they combined data from both blood microsamples and wearables to track lifestyle-associated changes in health.
This is part two of the blog series, in which we examine how the Stanford researchers developed a multi-omics assay that enabled them to measure more than 2,000 multi-omics biomarkers from just two 20 µL VAMS® tips on each Mitra® microsampling device. The measured analytes included polar metabolites, lipids, and proteins / peptides, as well as hormones and inflammatory biomarkers. 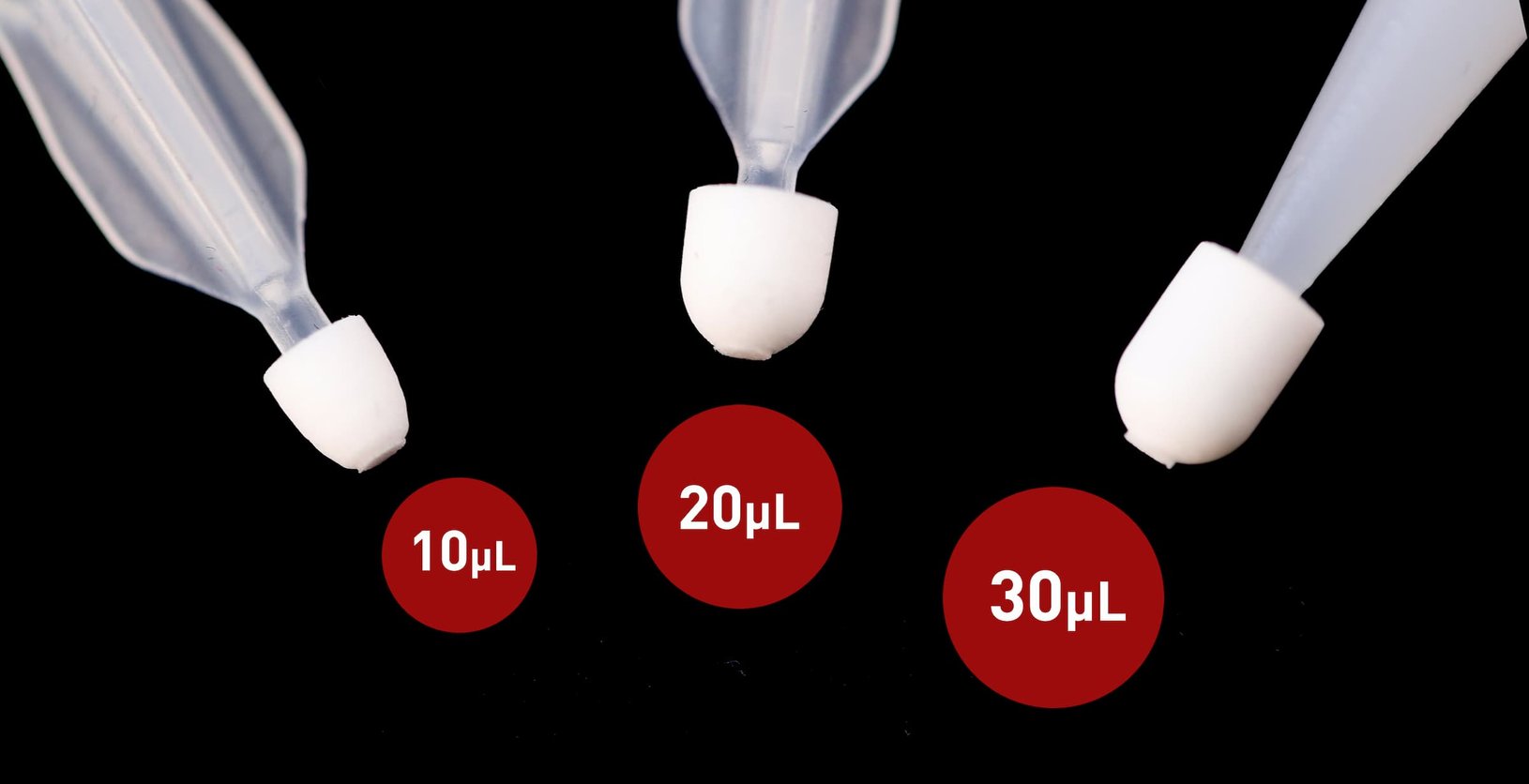
The Mitra® device with the original, patented VAMS® technology is available with a 10, 20 or 30 µL absorbent VAMS tip.
In this blog we also examine two of the Stanford group’s pilot studies that generated meaningful omics data. One study was a nutrition shake challenge among fasting study participants. The second was an in-depth multi-omics profiling study of data gathered from volunteers using wearables over a seven-day period.
Fluxomic Analysis or Metabolomic Flux Analysis
in Multi-Omics
The term fluxomics describes a number of different approaches to determine the rate of metabolic reactions, either through real-time monitoring (technically challenging) or through repeated and rapid blood sampling over a short time period (e.g., over several hours).
Although the Snyder et al study paper, published in a January 2023 issue of Nature Biomedical Engineering does not make reference to the term ‘fluxomics,’ the authors do mention ‘metabolomic flux analysis,’ which is essentially the same idea.
 One of the key concepts of omics analysis is that biomarker levels in the human body do not remain static. On the contrary, in an attempt to maintain homeostasis, different processes in the body are continually upregulated and down regulated. As a result, they are in a constant state of flux. This poses quite a technical challenge when trying to ascertain causality and predictability of specific processes.
One of the key concepts of omics analysis is that biomarker levels in the human body do not remain static. On the contrary, in an attempt to maintain homeostasis, different processes in the body are continually upregulated and down regulated. As a result, they are in a constant state of flux. This poses quite a technical challenge when trying to ascertain causality and predictability of specific processes.
 For example, is it possible that with continuous monitoring via a wearable to measure glucose levels that you can predict changes in c-peptide (diabetes biomarker) levels? Or do c-peptide levels also change as a result of natural biological rhythms, making prediction more difficult? It is questions like these that were addressed in the Snyder Lab’s pilot case studies, especially the second one.
For example, is it possible that with continuous monitoring via a wearable to measure glucose levels that you can predict changes in c-peptide (diabetes biomarker) levels? Or do c-peptide levels also change as a result of natural biological rhythms, making prediction more difficult? It is questions like these that were addressed in the Snyder Lab’s pilot case studies, especially the second one.
Multi-Omics Case Studies
Case Study 1: Phenotype responses to nutrition shake (Ensure®) consumption-
- Two Mitra-VAMS microsamples were collected from each of 28 subjects across 5 timepoints (0, 30, 60, 120 and 240 min).
- After the first collection point, all the subjects consumed a nutrition shake (Ensure) to see how the influx of nutritional contents impacted their biochemical pathways.
- Six metabolomic response metrics were created including carbohydrates, lipids, amino acids, insulin, FFA associated with sensitivity to insulin, and cytokines to monitor immune and inflammatory response.
- After processing the data, the researchers detected 768 analytes, including 560 metabolites, 155 lipids and 54 cytokines.
- Two Mitra-VAMS microsamples were collected from each of 28 subjects across 5 timepoints (0, 30, 60, 120 and 240 min).
-
- Using a Wilcoxon rank test, out of all the metabolites and lipids that were raised as a result of consuming the shake, most peaked at 60-120 min before returning to baseline at 240 min.
- According to an ANOVA test, 99 out of 560 metabolites, all 155 lipids, and 7 out of 54 cytokines had significantly changed. The researchers concluded, “These results demonstrate that multi-omics analysis from microsamples can be used to measure the metabolic response to [consuming an] Ensure shake.”
- Using fuzzy logic as a c-means clustering analysis, the researchers determined that three clusters across 5 timepoints behaved differently showing different patterns of kinetics.
- Unsurprisingly, Ensure shake components were detected in the blood (50%); out of 21 key metabolites detected, 17 were also included in the shake contents.
- Other observations were that both c-peptide and insulin (involved with glucose regulation) were increased as well as leptin (involved with a feeling of satiation).
- Using a Wilcoxon rank test, out of all the metabolites and lipids that were raised as a result of consuming the shake, most peaked at 60-120 min before returning to baseline at 240 min.
-
- Proinflammatory markers quickly decreased on consumption, indicating the shake may have anti-inflammatory properties.
- Although not statistically significant, there appeared to be a trend where those individuals with insulin resistance were in the group which responded faster to the effect of the shake.
- An enormous heterogeneity between individuals was seen.
- This highlighted the need for microsampling to monitor those with elevated LDLS and A1c, which the researchers indicated would be important for personalized future nutrition management.
- Proinflammatory markers quickly decreased on consumption, indicating the shake may have anti-inflammatory properties.
-
- Case Study 2: One volunteer wearing a Fitbit® smartwatch to continually measure pulse, steps and sleep, as well as a Dexcom glucose monitoring device, was regularly sampled with Mitra microsampling devices over 7 days.
- The Mitra sampling was as frequent as 30 minutes apart, so a total of 98 samples were collected.
- Over 2,000 analytes were detected across the same analyte categories as in the first study, resulting in over 200K biochemical measurements plus the continuous monitoring data.
- By alternating the amount of carbohydrate in the food on specific days, the omics data could reveal the concentration of the carbs consumed.
- As with the previous study, components found in a morning meal shake, were identified from the dried blood extracts.
- Case Study 2: One volunteer wearing a Fitbit® smartwatch to continually measure pulse, steps and sleep, as well as a Dexcom glucose monitoring device, was regularly sampled with Mitra microsampling devices over 7 days.
-
- When an analgesic was consumed over 4 days, the group could capture the pharmacokinetics (PK) of the drug, indicating a potential application for using this technique to personalize the PK of therapeutic drugs.
- The researchers were able to determine that the stress hormone cortisol didn’t seem to correlate with expected inter-day patterns, suggesting a requirement for higher frequency sampling.
- The researchers also determined that several proinflammatory cytokines were elevated over 3 days, which may have indicated an asymptomatic infection or other stressor. They highlighted the power of frequent sampling for early disease detection.
- The data indicated that different lipid classes showed distinct circadian patterns and were not driven by food consumption.
- In terms of using data from wearables to predict molecular changes: Out of 2,223 molecules that were detected, 447 (most of which were lipids) correlated to data collected from the continuous monitoring devices. For example, triglycerides were correlated to heart rate data.
- By further optimizing the data, the researchers showed that glucose correlated more with cytokines (thus immune response) and c-peptides. Blood coagulation pathways correlated with heart rate as well as other things.
- When an analgesic was consumed over 4 days, the group could capture the pharmacokinetics (PK) of the drug, indicating a potential application for using this technique to personalize the PK of therapeutic drugs.
-
- The researchers concluded that the causal relationships could be validated through future experiments.
Multi-Omics Study Authors’ Summary
- The studies showed the potential for large-scale comprehensive dynamic biomarker studies.
- The studies showed the potential of measuring multi-omics data using blood microsampling for personalized medicine and health, biomarker discovery, and therapeutic drug monitoring (TDM).
- More data could be collected if cellular DNA, RNA and epigenomes were also incorporated.
- A limitation of the second study was that it was only an n=1, so a larger cohort would show more generalized patterns.
- The researchers concluded, “Overall, we believe the multi-omics microsampling approach offers a promising opportunity to integrate with wearable data to improve precision healthcare.”
Neoteryx Comments
Imagine a future where a wearable detects a concerning change in a person’s heart rate and then advises the wearer to immediately collect a capillary blood sample to be mailed via standard dry shipping to a laboratory. Analysis in the lab for precision in temporal blood sampling may then enable early intervention and early disease detection, or a change in medication. This is just one example of how the multi-omics approaches being explored by researchers in Stanford’s Snyder Lab may be applied.
This work at Stanford University has demonstrated the power of combining wearables with volumetric blood microsampling for the analysis of a huge amount of useful data for precision healthcare.
In the ensuing years this technology will get more sophisticated, but there are still several hurdles to overcome. One hurdle includes figuring out how to manage such a huge volume of data from millions of individuals. Nevertheless, it is remarkable what can be measured from just a tiny drop of blood collected from a finger-prick and the potential it has for changing personalized medicine for the better.
This article was summarized for our readers by James Rudge, PhD, Neoteryx Technical Director. This is curated content. To learn more about the important research outlined in this blog, visit the original article in Nature Biomedical Engineering.

Access published, peer-reviewed papers on similar microsampling research studies in our Technical Resource Library.
Image Credits: iStock, Trajan Scientific and Medical
Share this
- Microsampling (206)
- Research, Remote Research (119)
- Venipuncture Alternative (105)
- Clinical Trials, Clinical Research (83)
- Mitra® Device (73)
- Therapeutic Drug Monitoring, TDM (51)
- Dried Blood Spot, DBS (39)
- Biomonitoring, Health, Wellness (30)
- Infectious Disease, Vaccines, COVID-19 (24)
- Blood Microsampling, Serology (23)
- Omics, Multi-Omics (21)
- Decentralized Clinical Trial (DCT) (20)
- Specimen Collection (18)
- Toxicology, Doping, Drug/Alcohol Monitoring, PEth (17)
- Skin Microsampling, Microbiopsy (14)
- hemaPEN® Device (13)
- Preclinical Research, Animal Studies (12)
- Pharmaceuticals, Drug Development (9)
- Harpera Device (7)
- Industry News, Microsampling News (5)
- Antibodies, MAbs (3)
- Company Press Release, Product Press Release (3)
- Environmental Toxins, Exposures (1)
- May 2026 (1)
- July 2025 (1)
- May 2025 (1)
- April 2025 (2)
- December 2024 (1)
- November 2024 (1)
- October 2024 (3)
- September 2024 (1)
- June 2024 (1)
- May 2024 (1)
- April 2024 (4)
- March 2024 (1)
- February 2024 (2)
- January 2024 (4)
- December 2023 (3)
- November 2023 (3)
- October 2023 (3)
- September 2023 (3)
- July 2023 (3)
- June 2023 (2)
- April 2023 (2)
- March 2023 (2)
- February 2023 (2)
- January 2023 (3)
- December 2022 (2)
- November 2022 (3)
- October 2022 (4)
- September 2022 (3)
- August 2022 (5)
- July 2022 (2)
- June 2022 (2)
- May 2022 (4)
- April 2022 (3)
- March 2022 (3)
- February 2022 (4)
- January 2022 (5)
- December 2021 (3)
- November 2021 (5)
- October 2021 (3)
- September 2021 (3)
- August 2021 (4)
- July 2021 (4)
- June 2021 (4)
- May 2021 (4)
- April 2021 (3)
- March 2021 (5)
- February 2021 (4)
- January 2021 (4)
- December 2020 (3)
- November 2020 (5)
- October 2020 (4)
- September 2020 (3)
- August 2020 (3)
- July 2020 (6)
- June 2020 (4)
- May 2020 (4)
- April 2020 (3)
- March 2020 (6)
- February 2020 (3)
- January 2020 (4)
- December 2019 (5)
- November 2019 (4)
- October 2019 (2)
- September 2019 (4)
- August 2019 (4)
- July 2019 (3)
- June 2019 (7)
- May 2019 (6)
- April 2019 (5)
- March 2019 (6)
- February 2019 (5)
- January 2019 (8)
- December 2018 (3)
- November 2018 (4)
- October 2018 (7)
- September 2018 (6)
- August 2018 (5)
- July 2018 (8)
- June 2018 (6)
- May 2018 (5)
- April 2018 (6)
- March 2018 (4)
- February 2018 (6)
- January 2018 (4)
- December 2017 (2)
- November 2017 (3)
- October 2017 (2)
- September 2017 (4)
- August 2017 (2)
- July 2017 (4)
- June 2017 (5)
- May 2017 (6)
- April 2017 (6)
- March 2017 (5)
- February 2017 (4)
- January 2017 (1)
- July 2016 (3)
- May 2016 (1)
- April 2016 (2)

No Comments Yet
Let us know what you think