Share this
low-resourced regions: microsampling makes research simpler
by Neoteryx Microsampling on Oct 14, 2019 5:07:00 AM
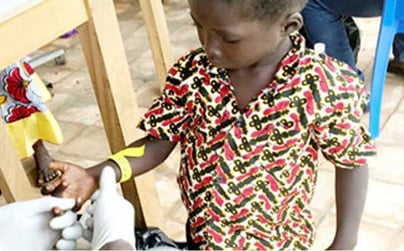
The Swiss Tropical and Public Health Institute is at the forefront of important research using volumetric absorptive microsampling technology with Mitra® microsampling devices in low-resourced regions, including locations in Africa.
Researchers from the Institute tested Mitra® devices based on VAMS® technology against more traditional Dried Blood Spot (DBS) cards in pediatric PK studies in rural Côte d’Ivoire.
Swiss TPH’s helminths team, including Dr. Daniela Hofmann, has performed work using Mitra® devices, including a study on evaluating different doses of albendazole to treat soil-transmitted helminths (STH).
"Albendazole is one of the very few medications listed on the WHO list of essential medicines to treat parasitic worm infections," says Dr. Hofmann. "In children, preventative chemotherapy, consisting of an annual or biannual single dose of albendazole at 400 mg, is recommended as a public health intervention where baseline prevalence of soil-transmitted infection is 20% or more."
To do her work, Dr. Hofmann travels to remote areas of the world to treat parasitic worms and takes blood sample donations to optimize future treatment for affected groups.
The study design is along similar lines to its earlier PK studies. "The research we are doing now follows an identical workflow as compared to earlier studies," says Hofmann. "Yet we are constantly interested in investigating the potential of novel and old anthelmintic drugs against different worms."
Once an LC-MS method is validated and a satisfactory correlation has been shown between samples obtained through venous blood sampling and dried blood microsampling, dose-finding PK studies in endemic regions (Cote d'Ivoire, in this case) are initiated.
"For a subgroup of the patients, finger-prick samples of blood were taken with Mitra® devices at defined time points," Dr. Hofmann says. "These samples lay the groundwork in evaluating and proposing improved doses and dosing regimens of albendazole in the treatment of STH."

Doing this sort of work in low-resourced regions poses unique challenges:
- Limited space and infrastructure: "Decreasing the total amount of material used, as well as being able to store blood samples in a simple and fast way, improves the workflow tremendously," says Dr. Hofmann. "Mitra® devices can be packed away immediately, do not require cooling or freezing, and are quite robust for handling and protected from dust, dirt, or spillover, and are thus very ideal."
- Low hygiene standards: "The absence of a clean facility or hospital increases the risk of infection doing venous blood sampling," Dr. Hofmann says. Here, too, VAMS® provides clear advantages.
- Language barriers: "It might happen that the study coordinators and nurses do not share a common language," says Dr. Hofmann. "It is important to have a simple tool to withdraw blood. The use of Mitra® devices can be illustrated and demonstrated easily and is very intuitive for most people."
With the use of Mitra with VAMS® in clinical trials, PK studies or therapeutic drug monitoring in low-resourced regions, "the whole analytical pipeline becomes more efficient and solid, and pharmacological studies that take place in marginalized areas become more feasible," Dr. Hofmann says. "What I like the most about using the Mitra® device is that it is very patient-friendly and nurse-friendly. It is easy and intuitive to handle and does not scare patients away from donating their blood."
Image Credits: Trajan, Neoteryx, iStock, Shutterstock
Share this
- Microsampling (206)
- Research, Remote Research (119)
- Venipuncture Alternative (105)
- Clinical Trials, Clinical Research (83)
- Mitra® Device (73)
- Therapeutic Drug Monitoring, TDM (51)
- Dried Blood Spot, DBS (39)
- Biomonitoring, Health, Wellness (30)
- Infectious Disease, Vaccines, COVID-19 (24)
- Blood Microsampling, Serology (23)
- Omics, Multi-Omics (21)
- Decentralized Clinical Trial (DCT) (20)
- Specimen Collection (18)
- Toxicology, Doping, Drug/Alcohol Monitoring, PEth (17)
- Skin Microsampling, Microbiopsy (14)
- hemaPEN® Device (13)
- Preclinical Research, Animal Studies (12)
- Pharmaceuticals, Drug Development (9)
- Harpera Device (7)
- Industry News, Microsampling News (5)
- Antibodies, MAbs (3)
- Company Press Release, Product Press Release (3)
- Environmental Toxins, Exposures (1)
- July 2025 (1)
- May 2025 (1)
- April 2025 (2)
- December 2024 (2)
- November 2024 (1)
- October 2024 (3)
- September 2024 (1)
- June 2024 (1)
- May 2024 (1)
- April 2024 (4)
- March 2024 (1)
- February 2024 (2)
- January 2024 (4)
- December 2023 (3)
- November 2023 (3)
- October 2023 (3)
- September 2023 (3)
- July 2023 (3)
- June 2023 (2)
- April 2023 (2)
- March 2023 (2)
- February 2023 (2)
- January 2023 (3)
- December 2022 (2)
- November 2022 (3)
- October 2022 (4)
- September 2022 (3)
- August 2022 (5)
- July 2022 (2)
- June 2022 (2)
- May 2022 (4)
- April 2022 (3)
- March 2022 (3)
- February 2022 (4)
- January 2022 (5)
- December 2021 (3)
- November 2021 (5)
- October 2021 (3)
- September 2021 (3)
- August 2021 (4)
- July 2021 (4)
- June 2021 (4)
- May 2021 (4)
- April 2021 (3)
- March 2021 (5)
- February 2021 (4)
- January 2021 (4)
- December 2020 (3)
- November 2020 (5)
- October 2020 (4)
- September 2020 (3)
- August 2020 (3)
- July 2020 (6)
- June 2020 (4)
- May 2020 (4)
- April 2020 (3)
- March 2020 (6)
- February 2020 (3)
- January 2020 (4)
- December 2019 (5)
- November 2019 (4)
- October 2019 (2)
- September 2019 (4)
- August 2019 (4)
- July 2019 (3)
- June 2019 (7)
- May 2019 (6)
- April 2019 (5)
- March 2019 (6)
- February 2019 (5)
- January 2019 (8)
- December 2018 (3)
- November 2018 (4)
- October 2018 (7)
- September 2018 (6)
- August 2018 (5)
- July 2018 (8)
- June 2018 (6)
- May 2018 (5)
- April 2018 (6)
- March 2018 (4)
- February 2018 (6)
- January 2018 (4)
- December 2017 (2)
- November 2017 (3)
- October 2017 (2)
- September 2017 (4)
- August 2017 (2)
- July 2017 (4)
- June 2017 (5)
- May 2017 (6)
- April 2017 (6)
- March 2017 (5)
- February 2017 (4)
- January 2017 (1)
- July 2016 (3)
- May 2016 (1)
- April 2016 (2)



No Comments Yet
Let us know what you think