Share this
Expert Q&A: minimally invasive sampling of skin diseases
by Florian Lapierre, PhD, Microsampling Product Director on Feb 6, 2023 9:00:00 AM
We are exploring the different ways that researchers apply the Neoteryx Harpera™ from Trajan, a minimally invasive skin biopsy device. Our Microsampling Product Director, Florian Lapierre, PhD, recently spoke with Assistant Professor Wim Adriaensen (Clinical Immunology Unit), Dr. Kristien Cloots (Unit of Neglected Tropical Disease-Public Health) and Dr. Saskia van Henten (Unit of Neglected Tropical Diseases-Clinical Sciences) at the Institute of Tropical Medicine (ITM).
The ITM in Antwerp, Belgium is an internationally renowned institute that provides education, research and services in tropical medicine and health care in developing countries.
Coming from different disciplines, the three researchers are working together to study the value of the Harpera™ Microbiopsy™ Punch in research on the tropical skin disease Leishmaniasis in Africa and India.

Image: ITM Researchers from Left to Right: Wim Adriaensen, Kristien Cloots, Saskia van Henten
To learn how researchers are collecting skin biopsies to identify and monitor skin diseases in remote locations, read the Q&A below:
Dr. Lapierre: Thank you for speaking with me about your work! For my first question, I'd like to ask what motivated you and the team to choose careers in the field of infectious disease monitoring and public health research?
Assist. Prof. Adriaensen: We were strongly driven by the clinical and public health impact one can have in this field, in particular in low- and middle-income countries where good quality evidence can lead to immediate improvements in standard of care, and where there is a need for global-coordinated efforts in control of these diseases.
Q: We are learning that compliance can be an issue when conducting tropical disease monitoring to support eradication. Why do you think compliance is a challenge for communities affected by such diseases?
Dr. Cloots: Diagnosis of many neglected skin tropical diseases, like cutaneous leishmaniasis or leprosy, is based on rather invasive skin sample collection for microscopy and pathogen detection, often accompanied by bleeding after sample collection.
This contributes to patients' reluctance to travel far to specialized clinics for diagnosis. In practice, most patients in the countries we work in present to the hospital late with large and severe lesions.
However, early diagnosis and treatment could prevent scar formation, stigmatization and could be important for disease control.
Therefore, minimally invasive tools that can be applied by healthcare workers in the communities would be highly warranted to achieve early detection and disease control.
Q: Trajan’s Harpera™ Microbiopsy™ Punch is designed to collect only the necessary number of cells required for analytical testing in order to ensure high-quality results and improve patient compliance. What was your experience of this when you used Harpera™ Microbiopsy™ Punch in your study? Did the tool provide sufficient biological material to be clinically relevant compared to other industry tools or practices?
Dr. van Henten: At ITM, we first started using this Microbiopsy device in two small proof-of-concept studies. Firstly, we wanted to see how well the device worked in Ethiopia compared to the routine sampling method for cutaneous leishmaniasis called skin slit sampling.
Because the Microbiopsy takes a much smaller amount of tissue, we were expecting it to be inferior compared to the routine sample, but to our surprise, more patients tested positive with the Microbiopsy sample despite many having crusted lesions!
This shows that more tissue does not automatically mean a better sample. In fact, it seems that Harpera is able to reach the high parasite niche of the skin and takes a sample that has a good ratio of human and parasite DNA.
Another benefit of Harpera is that it takes a consistent amount of sample, while with other available sampling techniques it is hard to standardize the sample quantity.
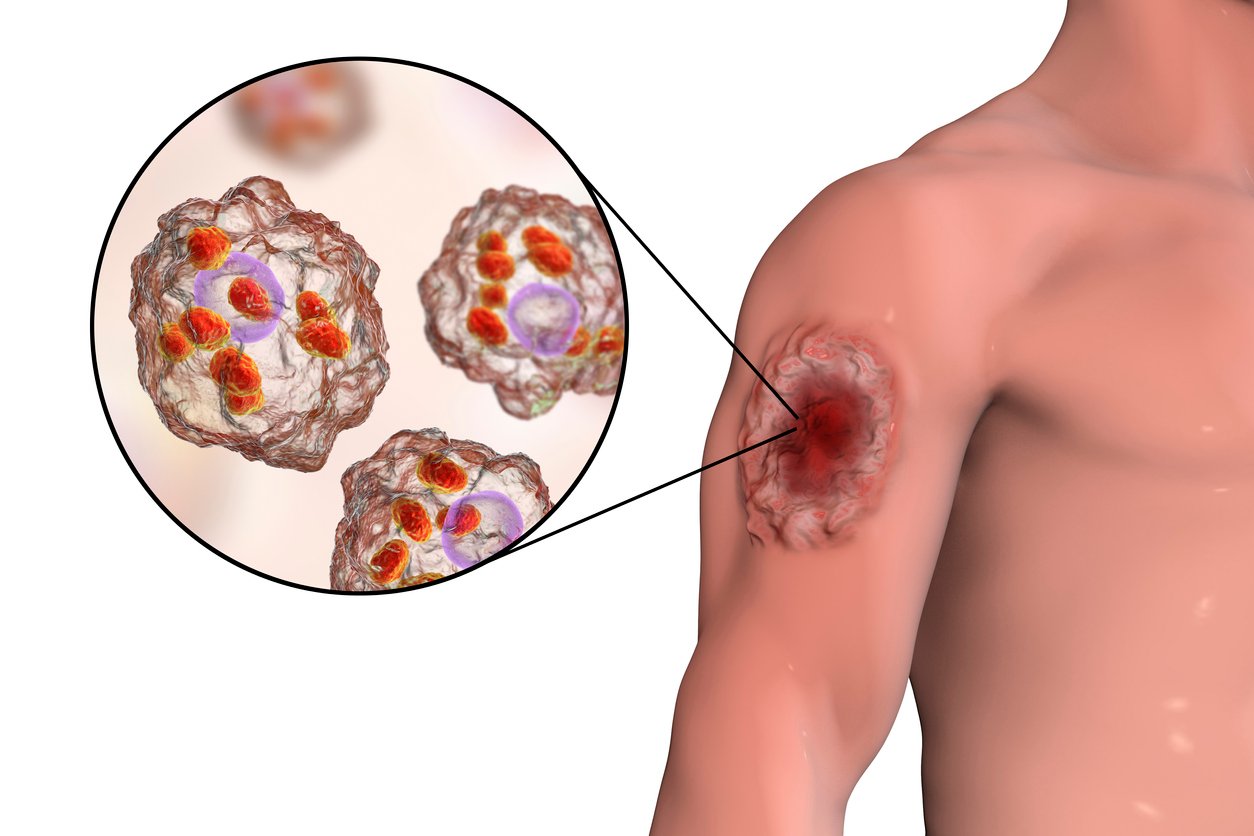
Image: Cutaneous leishmaniasis ulcer and close-up view of Leishmania amastigotes
Dr. Cloots: In India, we showed that the Harpera device could pick up Leishmania parasites in visceral leishmaniasis and post-kala-azar dermal leishmaniasis (PKDL) patients, as well as in participants with asymptomatic infection with the Leishmania parasite responsible for causing the deadly visceral form of disease.
This indicates that the Microbiopsies could be used for diagnosis of PKDL. It also shows potential to be used for studying transmission potential of the feeding sand flies that act as the vector for the disease, as it mimics the bite of a sandfly.
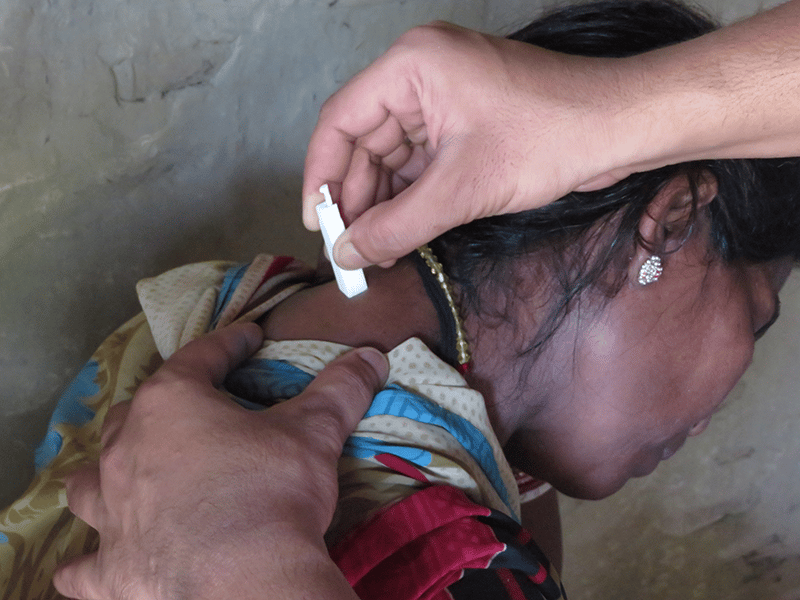
Image: The Harpera device being applied for the study in the field
Q: Trajan’s Harpera™ Microbiopsy™ Punch is designed to minimize the invasiveness of a biopsy and simplify the procedure of sampling. What were your key findings of the Harpera’s overall performance during your study?
Assist. Prof. Adriaensen: Cutaneous leishmaniasis patients experienced this Microbiopsy sampling device as much less painful than the skin slit sample, which indicates that Microbiopsy samples could be taken without local anesthesia with only minimal inconvenience to patients.
In contrast to other types of sampling, the Microbiopsy does not lead to bleeding, which makes it more comfortable for patients as well as the staff taking the samples. This convinced us to incorporate the Microbiopsy device in more of our planned diagnostic and transmission studies.
Q: What did you achieve using Harpera that you wouldn’t have been able to achieve through other sampling methods?
Dr. van Henten: The minimally invasive and standardized sample collection with Harpera allows us to sample patients and their lesions multiple times and monitor the effect of different treatments on the pathogen load over time.
The minimal burden of collection also allows us to study the pathogen load within detailed outer and inner regions of the lesions. We also believe the Microbiopsies to be less operator-dependent, enabling data merging of multiple study sites.

Image: Close-Up of the Harpera™ microbiopsy device in action
Q: What is your advice to others who would like to implement Trajan’s Microbiopsy device in their field?
Dr. Cloots: The Harpera™ Microbiopsy™ Punch has a true potential to simplify diagnostic and surveillance efforts for many vector-borne or skin-tropic diseases.
Q: What are your upcoming projects involving the use of Trajan’s Microbiopsy device?
Dr. van Henten: We are continuing to collect evidence of its diagnostic accuracy in cutaneous leishmaniasis in different settings and lesion characteristics (e.g., crusted, ulcerated, nodular, etc.), but also in other skin NTDs to facilitate its implementation.
In addition, we are implementing its use in ongoing treatment clinical trials to monitor parasite load reduction and location within the lesion, or to study vaccine-induced skin-resident immunity in vaccination studies.
Q: Where do you predict person-centered technology will be in five years?
Assist. Prof. Adriaensen: We believe there is a great need for patient-centric or person-centered technology (e.g., tele-dermatology, self-sampling) in infectious diseases to, for instance, better reach rural communities located far from healthcare facilities.
Such technologies would also enable remote self-testing in case of highly contagious pathogens, which would help to reduce healthcare worker risks.
For more insights from the ITM research group using the Harpera Microbiopsy device to study cutaneous leishmaniasis, watch this video presentation by Dr. Saskia van Henten on YouTube: https://www.youtube.com/watch?v=9Bt_O3nox8A
Image Credits: Trajan, iStock, Abiy Ayele
Share this
- Microsampling (206)
- Research, Remote Research (119)
- Venipuncture Alternative (105)
- Clinical Trials, Clinical Research (83)
- Mitra® Device (73)
- Therapeutic Drug Monitoring, TDM (51)
- Dried Blood Spot, DBS (39)
- Biomonitoring, Health, Wellness (30)
- Infectious Disease, Vaccines, COVID-19 (24)
- Blood Microsampling, Serology (23)
- Omics, Multi-Omics (21)
- Decentralized Clinical Trial (DCT) (20)
- Specimen Collection (18)
- Toxicology, Doping, Drug/Alcohol Monitoring, PEth (17)
- Skin Microsampling, Microbiopsy (14)
- hemaPEN® Device (13)
- Preclinical Research, Animal Studies (12)
- Pharmaceuticals, Drug Development (9)
- Harpera Device (7)
- Industry News, Microsampling News (5)
- Antibodies, MAbs (3)
- Company Press Release, Product Press Release (3)
- Environmental Toxins, Exposures (1)
- May 2026 (1)
- July 2025 (1)
- May 2025 (1)
- April 2025 (2)
- December 2024 (1)
- November 2024 (1)
- October 2024 (3)
- September 2024 (1)
- June 2024 (1)
- May 2024 (1)
- April 2024 (4)
- March 2024 (1)
- February 2024 (2)
- January 2024 (4)
- December 2023 (3)
- November 2023 (3)
- October 2023 (3)
- September 2023 (3)
- July 2023 (3)
- June 2023 (2)
- April 2023 (2)
- March 2023 (2)
- February 2023 (2)
- January 2023 (3)
- December 2022 (2)
- November 2022 (3)
- October 2022 (4)
- September 2022 (3)
- August 2022 (5)
- July 2022 (2)
- June 2022 (2)
- May 2022 (4)
- April 2022 (3)
- March 2022 (3)
- February 2022 (4)
- January 2022 (5)
- December 2021 (3)
- November 2021 (5)
- October 2021 (3)
- September 2021 (3)
- August 2021 (4)
- July 2021 (4)
- June 2021 (4)
- May 2021 (4)
- April 2021 (3)
- March 2021 (5)
- February 2021 (4)
- January 2021 (4)
- December 2020 (3)
- November 2020 (5)
- October 2020 (4)
- September 2020 (3)
- August 2020 (3)
- July 2020 (6)
- June 2020 (4)
- May 2020 (4)
- April 2020 (3)
- March 2020 (6)
- February 2020 (3)
- January 2020 (4)
- December 2019 (5)
- November 2019 (4)
- October 2019 (2)
- September 2019 (4)
- August 2019 (4)
- July 2019 (3)
- June 2019 (7)
- May 2019 (6)
- April 2019 (5)
- March 2019 (6)
- February 2019 (5)
- January 2019 (8)
- December 2018 (3)
- November 2018 (4)
- October 2018 (7)
- September 2018 (6)
- August 2018 (5)
- July 2018 (8)
- June 2018 (6)
- May 2018 (5)
- April 2018 (6)
- March 2018 (4)
- February 2018 (6)
- January 2018 (4)
- December 2017 (2)
- November 2017 (3)
- October 2017 (2)
- September 2017 (4)
- August 2017 (2)
- July 2017 (4)
- June 2017 (5)
- May 2017 (6)
- April 2017 (6)
- March 2017 (5)
- February 2017 (4)
- January 2017 (1)
- July 2016 (3)
- May 2016 (1)
- April 2016 (2)



No Comments Yet
Let us know what you think